Expanded Display Interface
Larger, high-visibility screen for improved readability.
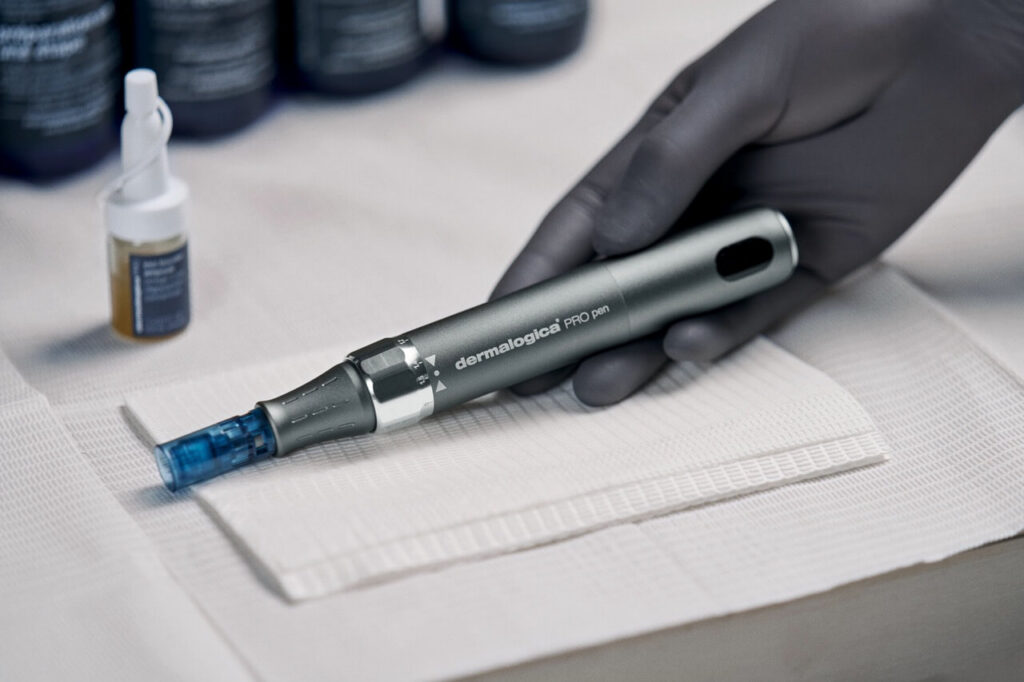
Pro Pen is an FDA 510(k) cleared class II medical device and is intended to be used as a treatment to improve the appearance of facial acne scars in adults age 22 years and older. Additionally, we have obtained ISO 13485 Certification, cementing Dermalogica as a medical device manufacturer by international standard.
Together, they place the Pro Pen in a top tier of microneedling devices globally – defined by regulation and scientific review.
Larger, high-visibility screen for improved readability.
Battery or cable operated to adjust to every environment. 4-hour battery charge.
Durable housing engineered for professional use.
From 6,300 to 7,700 RPM for customized treatments.
Fine-tune needle penetration depth for customized treatment results.
Contoured grip to help support accurate handing throughout treatment session.
The new 14-pin cartridge has been intentionally designed to deliver next-level results through smarter engineering. The 14-pin layout allows for evenly distributed microchannels, which supports effective collagen stimulation while maintaining control and comfort during treatment.


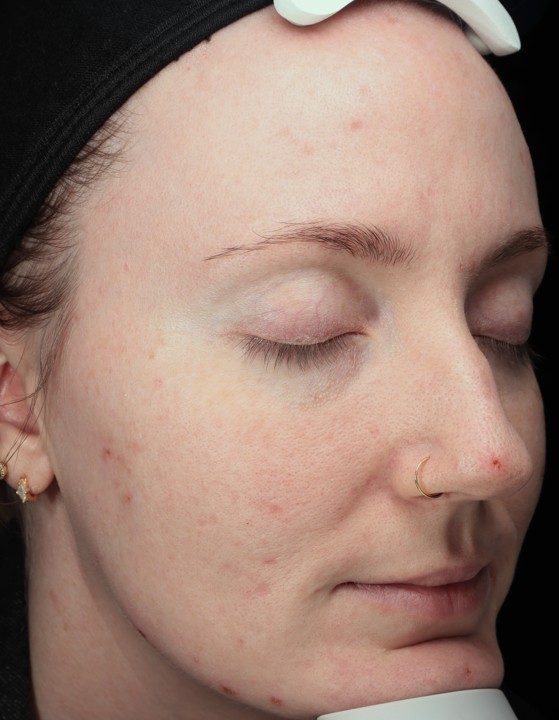
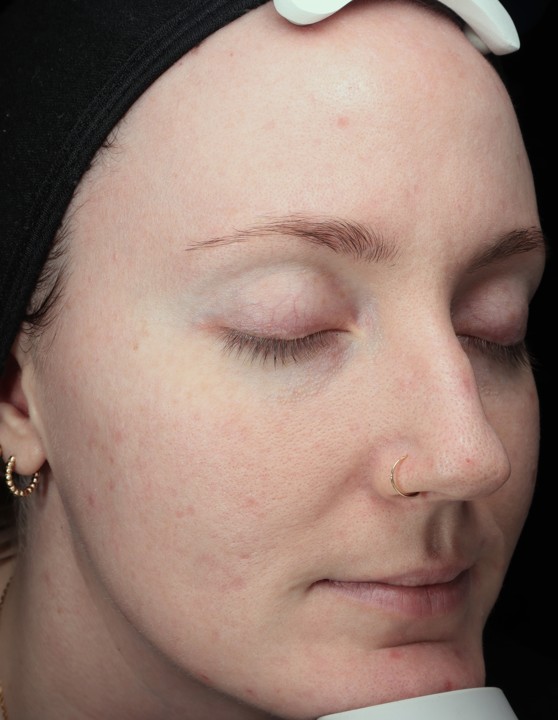


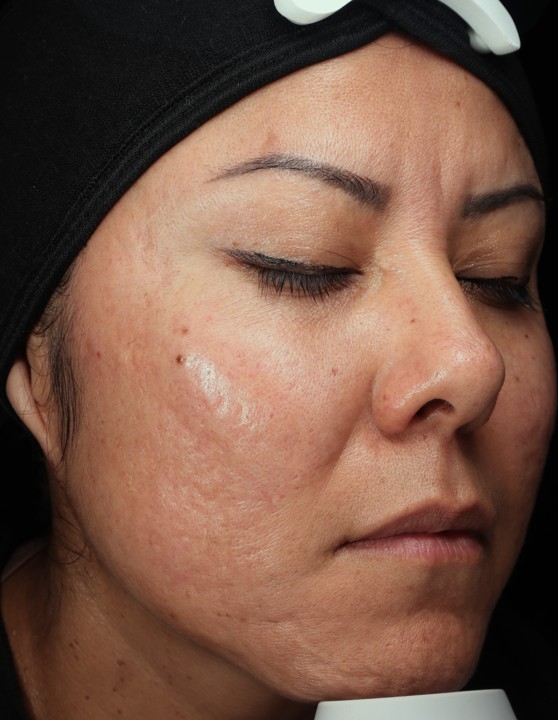
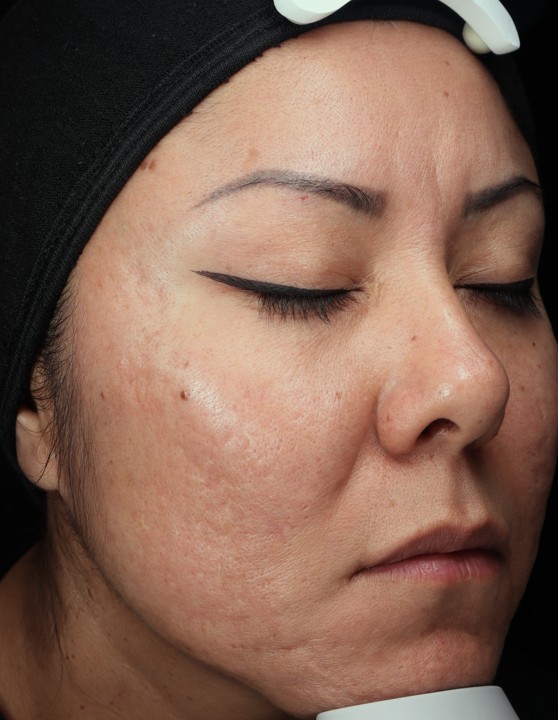
Bring the Dermalogica Pro Pen Microneedling system into your business today and start bridging the gap between clinical results and patient trust. Connect with a business development manager to get started.
Learn how the Pro Pen can support our non-medical nano-infusion technology, a modality created with scope of mind and patient demand in mind.